Aetna to Pay $117.7M to Settle Medicare Fraud Claims





Washington D.C. - March 12th, 2026 - CVS Health's Aetna unit has agreed to a $117.7 million settlement with the U.S. government to resolve claims of defrauding Medicare through improper billing practices for pharmaceutical services. This settlement, announced earlier this week, is not an isolated incident, but rather a bellwether for growing federal and public scrutiny of Pharmacy Benefit Managers (PBMs) and their impact on healthcare costs.
The Justice Department alleges that between 2011 and 2019, Corbit Health Solutions (acquired by Aetna in 2011) and its successor, Aetna Inc., knowingly submitted false claims for reimbursement under Medicare Part D. The core of the alleged fraud centered on artificially inflating the cost of prescription drugs and exploiting loopholes within the Medicare system. Specifically, investigators found that Corbit/Aetna falsely reported drugs as being dispensed through retail pharmacies when, in reality, they were being fulfilled via mail-order pharmacies. This misrepresentation allowed the companies to receive higher reimbursement rates, effectively siphoning funds from a program designed to provide affordable healthcare for seniors.
Attorney General Merrick Garland, in a statement released alongside the announcement, emphasized the seriousness of the offense. "Today's settlement holds Aetna accountable for its role in defrauding Medicare and exploiting a vital program for seniors," Garland stated. "The Justice Department will continue to vigorously pursue those who seek to profit illegally from taxpayer-funded healthcare programs."
CVS Health, while acknowledging the settlement, maintains that it has since strengthened its compliance programs to prevent similar occurrences. However, this assertion comes amidst a broader national conversation about the lack of transparency within the PBM industry and the potential for systematic abuse.
The Rise of PBMs and Growing Concerns
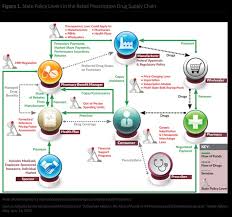
PBMs like Aetna (through its various subsidiaries) act as intermediaries between drug manufacturers, insurance companies, and pharmacies. They negotiate drug prices, create formularies (lists of covered drugs), and process claims. While initially intended to control costs and improve efficiency, PBMs have become increasingly complex, and critics argue that their business model incentivizes maximizing profits rather than prioritizing patient affordability.
The core issue lies in a lack of transparency. PBMs often receive rebates and discounts from drug manufacturers, but the extent to which these savings are passed on to consumers - or even to insurance companies - is often opaque. This lack of clarity allows PBMs to potentially pocket a significant portion of the savings, driving up overall healthcare costs.
Recent Legislative Efforts and Future Outlook
The Aetna settlement adds fuel to a growing legislative push for greater PBM regulation. Several states are considering or have already passed laws requiring PBMs to disclose more information about their pricing practices and rebate arrangements. At the federal level, there's bipartisan support for similar reforms. A bill gaining traction in Congress, the "Improving Transparency and Lowering Costs in Prescription Drugs Act," would mandate PBMs to publicly report data on rebates, discounts, and administrative fees.
Industry analysts predict that further legal action against PBMs is likely. The Federal Trade Commission (FTC) is currently conducting a comprehensive investigation into the PBM industry, focusing on potential anti-competitive practices and conflicts of interest. The findings of this investigation, expected to be released later this year, could pave the way for even more stringent regulations and enforcement actions.
"We are entering a new era of PBM accountability," says Dr. Eleanor Vance, a health policy expert at the National Institute for Health Reform. "The Aetna settlement is just the tip of the iceberg. The public is demanding greater transparency, and policymakers are responding. PBMs will need to adapt to a more regulated environment or risk facing further legal and financial consequences."
The impact of these changes will be far-reaching. Increased transparency could lead to lower drug prices, improved patient access to affordable medications, and a more sustainable healthcare system overall. However, PBMs argue that increased regulation could stifle innovation and limit their ability to negotiate effectively with drug manufacturers. The coming years will be crucial in determining how the evolving landscape of PBM regulation will reshape the future of prescription drug access and affordability in the United States.
Read the Full U.S. News & World Report Article at:
https://www.usnews.com/news/top-news/articles/2026-03-11/cvs-aetna-pays-117-7-million-to-settle-us-claims-it-defrauded-medicare
on: Tue, Mar 03rd
by: WTOP News

on: Mon, Mar 02nd
by: Press-Telegram

on: Sun, Mar 01st
by: Orlando Sentinel

on: Thu, Feb 12th
by: Dallas Express Media
Colony Ridge Settlement: DOJ Scrutiny Over Lending Practices

on: Fri, Feb 06th
by: Yahoo News

on: Mon, Feb 02nd
by: The Gazette

on: Thu, Jan 29th
by: CNN

on: Tue, Jan 20th
by: Politico
on: Wed, Jan 14th
by: The Messenger

on: Tue, Dec 23rd 2025
by: Atlanta Journal-Constitution

on: Thu, Nov 20th 2025
by: Associated Press
Trump-era Health Subsidies to End in 2025: Premiums Set to Rise

on: Tue, Nov 18th 2025
by: Politico
White-House-Backed Health Bill Aims to Cut Drug Costs and Shift Global Trade Dynamics
















































